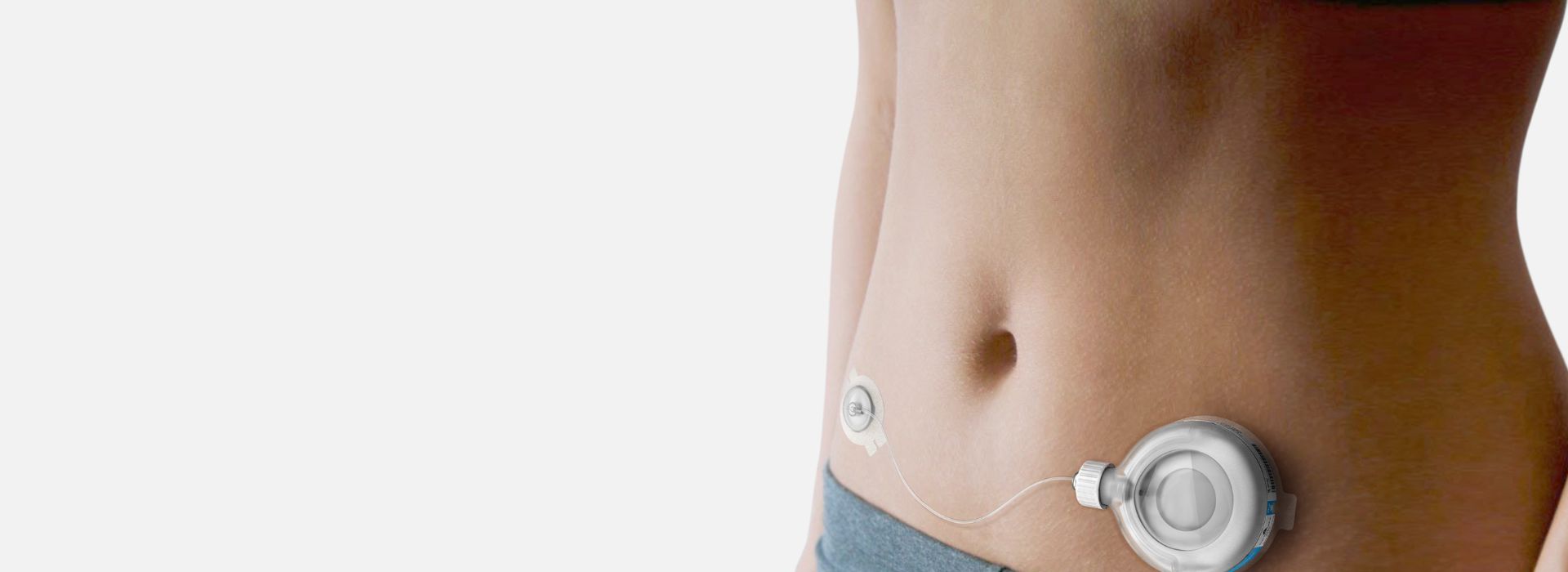
Drug delivery
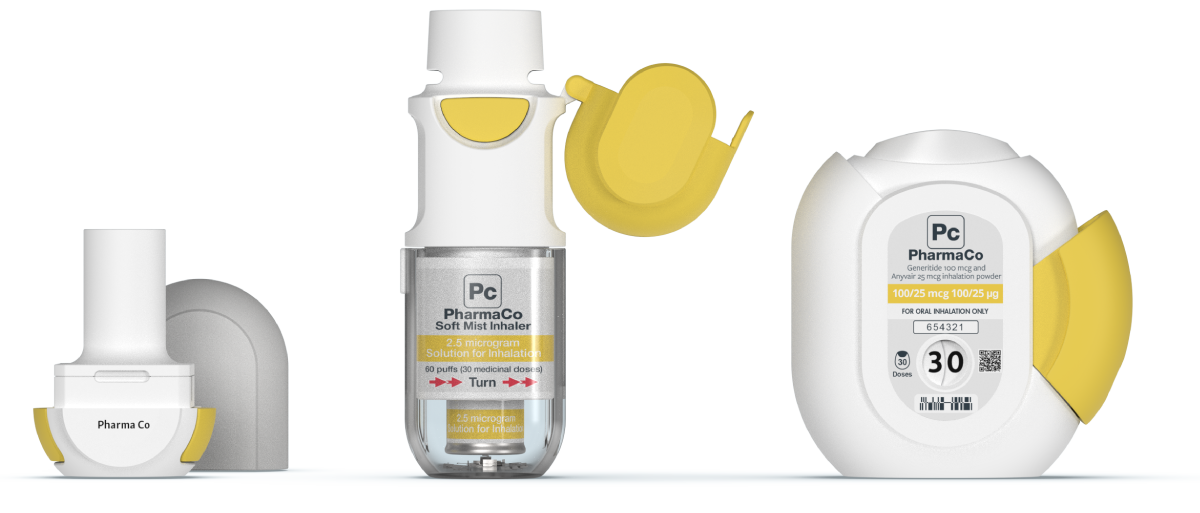
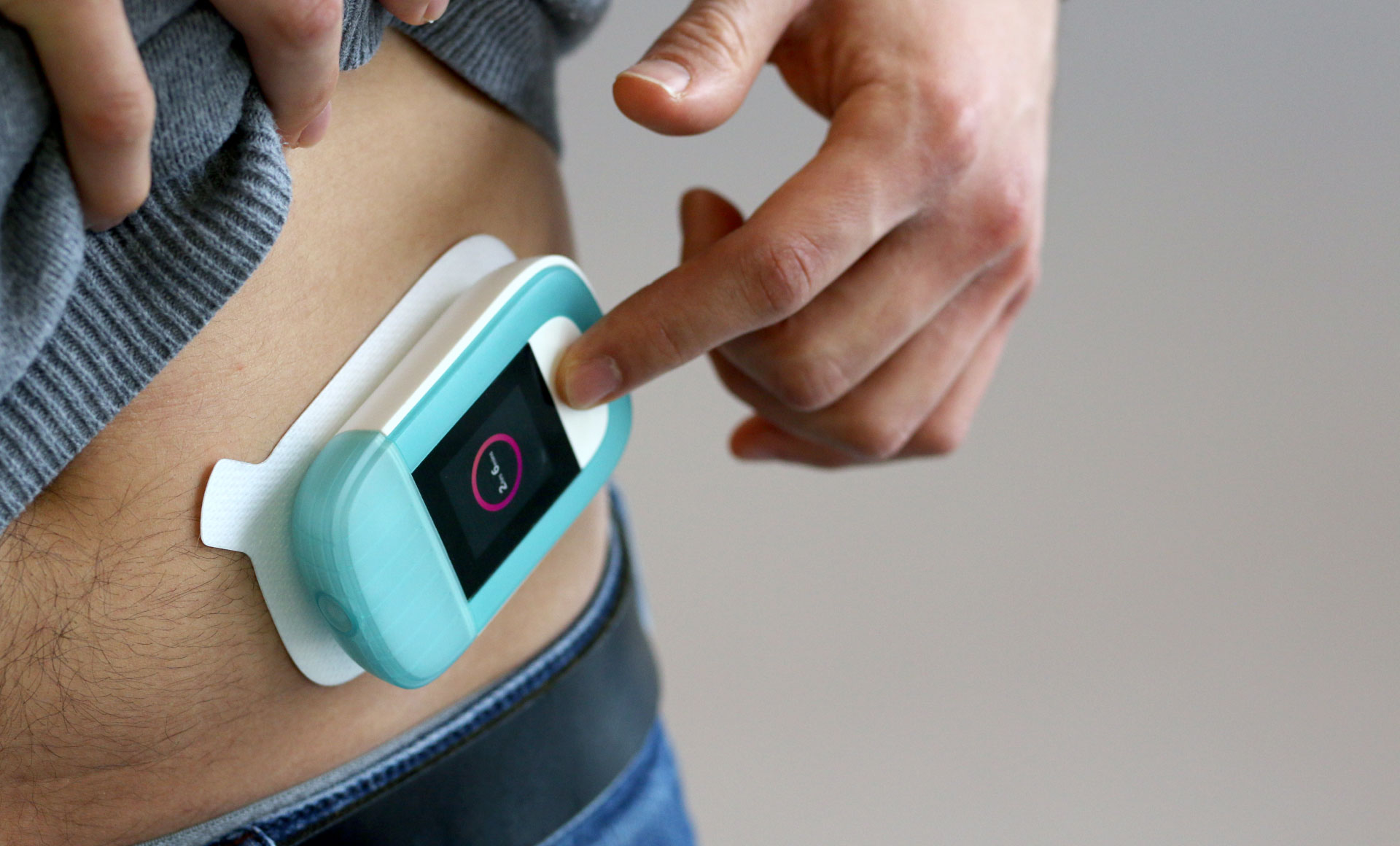
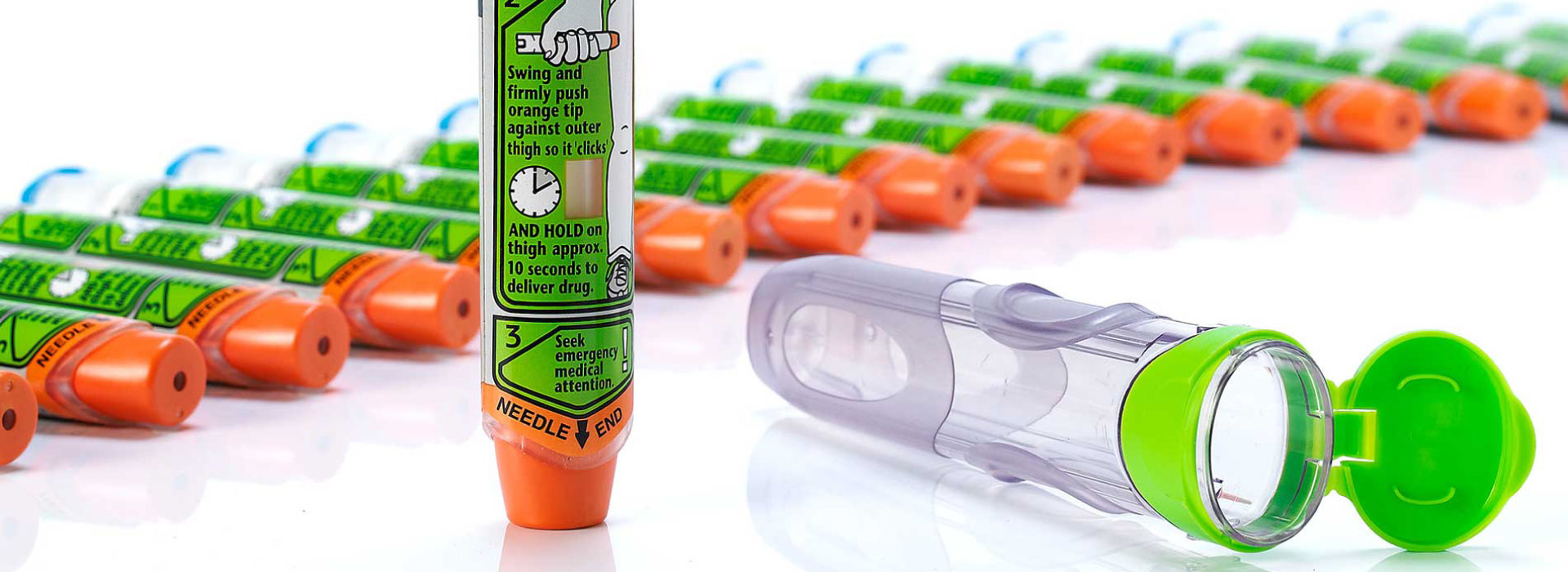
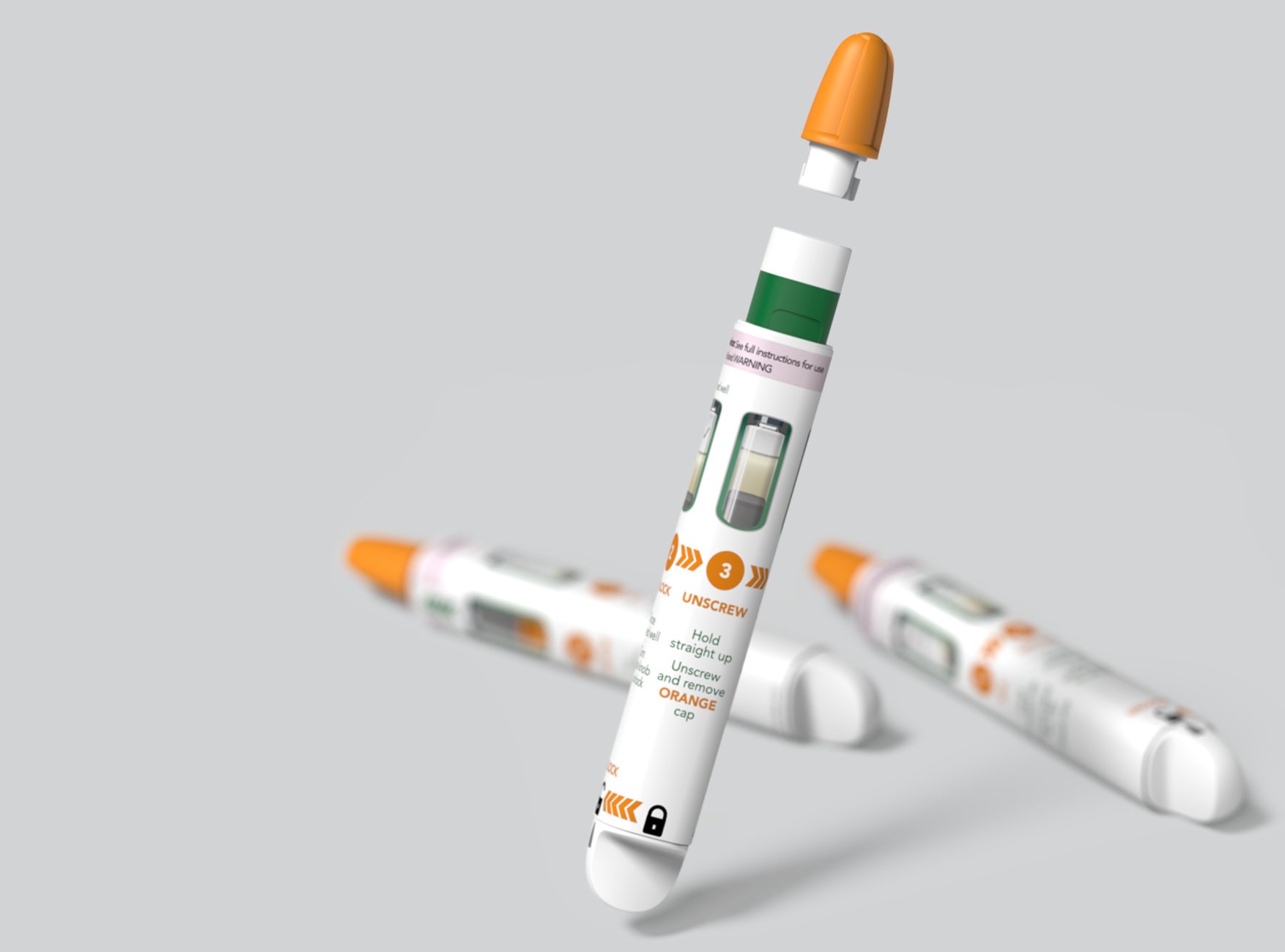
With over 35 years of expertise developing drug delivery systems and combination products, we work with you to bridge the gap between the user and the technology to deliver creative solutions for your therapeutic area. Our drug delivery device expertise covers everything from developing injection and respiratory devices through to emergency use devices, implantable technologies, innovative drug delivery systems for surgical applications, connected drug delivery systems and so much more.